
SOLVED: Aqueous solutions of sodium bicarbonate and sulfuric acid react to produce carbon dioxide according to the following equation: 2 NaHCO3(aq) + H,SO4(aq) = 2 C02(g) + Na2 SO4(aq) +2H20(1) If 106.6
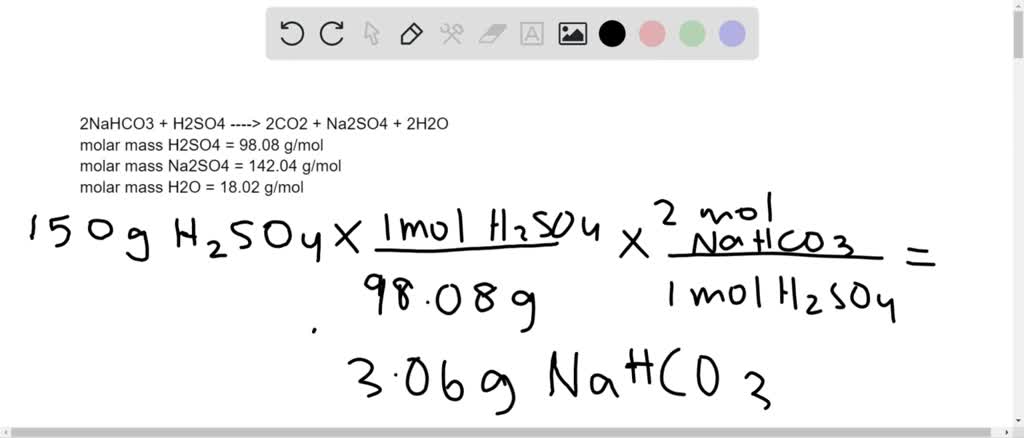
SOLVED:In a soda-acid fire extinguisher, concentrated sulfuric acid reacts with sodium hydrogen carbonate to produce carbon dioxide, sodium sulfate, and water. a. How many moles of sodium hydrogen carbonate would be needed
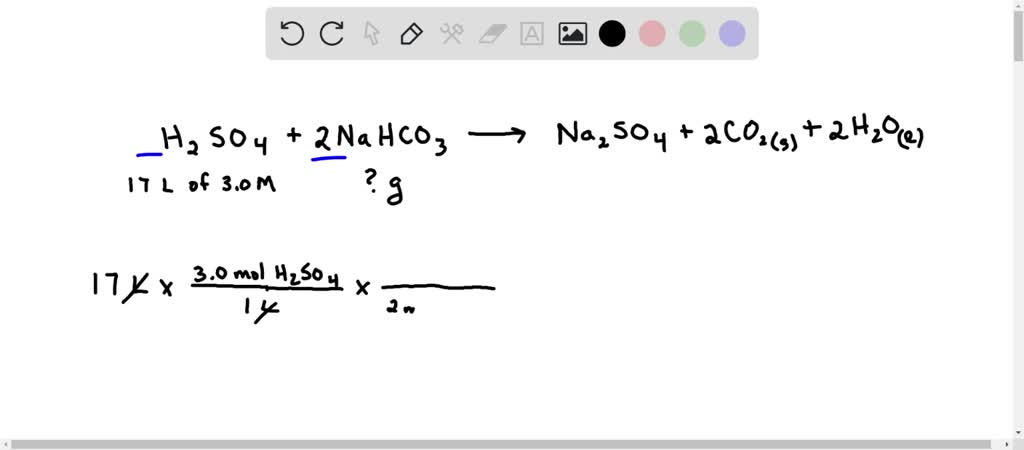
SOLVED: Students working in lab accidentally spilled 17 L of 3.0 M H2SO4 solution. They find a large container of acid neutralizer that contains baking soda, NaHCO3. How many grams of baking

Question Video: Determining the Concentration of Sulfuric Acid Via Titration with Sodium Carbonate | Nagwa

SOLVED: Aqueous solutions of sodium bicarbonate and sulfuric acid react to produce carbon dioxide according to the following equation: 2 NaHCOs(aq) + H,SO4(aq) 3 2CO2(g) + NazSO4(aq) + 2 HzO() If 105.4
















